
The TGA is reviewing all COVID-19-specific laboratory antigen tests and rapid antigen tests (RATs), including point-of-care and self-tests, included in the Australian Register of Therapeutic Goods (ARTG). The purpose of the review is to determine if tests have been impacted by the current known variants of SARS-CoV-2, with ongoing activity to assess their ability to detect emerging variants.
Clongene Antigen Rapid Tests (POCT and self-testing are equivalent) have passed the independent verification of TGA, and can detect both Delta and Omicron variants. The LoD meets the requirements of WHO guidelines (within the WHO guidelines of 100-1000 TCID50/mL).
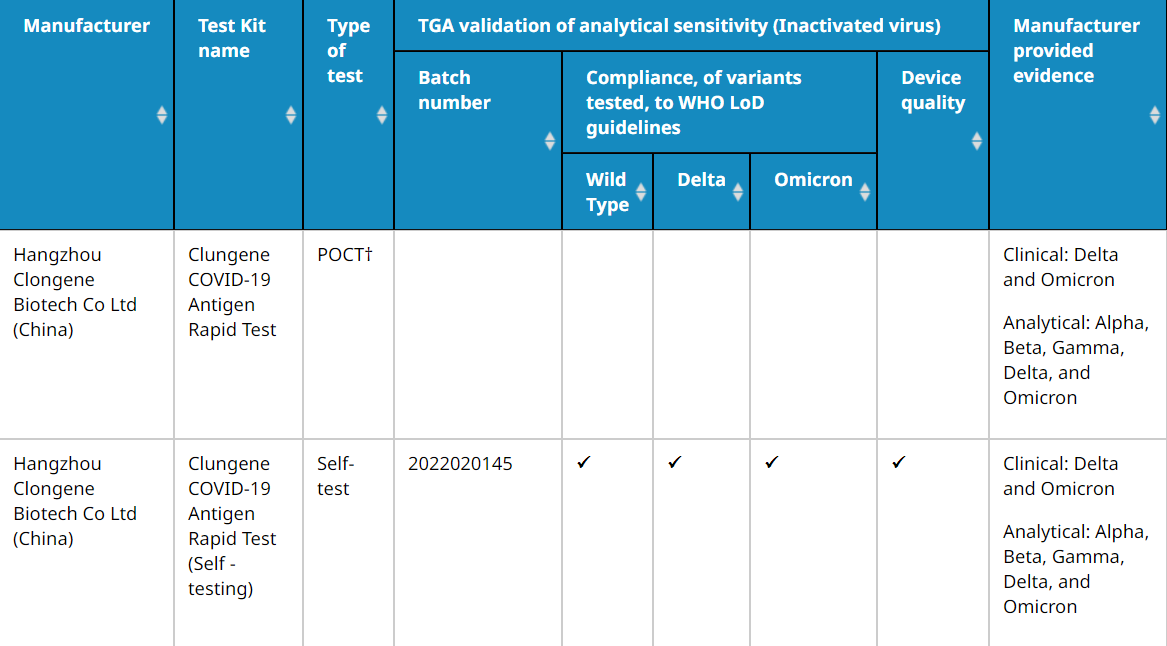
(More information click here.)
As an international supplier of IVD diagnostic reagents, we will continue to fight for international health and global epidemic prevention, and provide our customers with more high-quality products and services.